December 27, 2022
J Endod Microsurg. 2023;2: 24-33.
Under a Creative Commons license
HOW TO CITE THIS ARTICLE (AMA Referencing)
Floratos S, Molonis V, Tsolakis A, Kykalos S, Kontzoglou K. Bone window technique in endodontic microsurgery – report of two cases. J Endod Microsurg. 2023;2:24-33. https://doi.org/10.23999/jem.2023.2.3
Contents: Abstract | Key Words | Introduction | Case Reports | Discussion | Conclusions | Conflict of interest | References (17)
NATIONAL REPOSITORY OF ACADEMIC TEXTS
https://nrat.ukrintei.ua/en/searchdoc/2023U000305/
ABSTRACT
Endodontic microsurgery was introduced in the ’90s and has significantly increased the success rate of apical surgical intervention the last few decades. Utilizing the dental operating microscope, ultrasonic tips for root end preparation and biocompatible root end filling materials, predictably manages the apical pathology preserving the buccal cortical plate. The bone window technique for buccal approach to the apical area involves the use of piezoelectric unit to prepare and elevate a buccal cortical bony window and the reposition of the bone after the apical root end filling is completed. Two cases are reported in this article, highlighting the importance of endodontic microsurgery and buccal bone window technique in addressing apical pathology in a minimally invasive way, preserving the hard tissues and the tooth structure. Cases were reevaluated clinically and radiographically after a period of 3 months up to 36 months.
KEY WORDS
Bone window; piezoelectric surgery; autologous graft; endodontic microsurgery; root resection, apicoectomy; ultrasonics
INTRODUCTION
Endodontic microsurgical approach was introduced in 1990s and today is enormously advanced. The success rates rise to 92% [1], which proves that it is a predictable treatment method. The advantages derive from its main components including the dental operating microscope, the use of ultrasonic tips for root end preparation coaxial to the canal and more biocompatible root end filling materials. Inspection under high magnification is the key stage of microsurgery that is missing from the traditional surgical technique [2, 3]. A careful inspection identifies the possible reasons for failure of the nonsurgical treatment. Due to the improved visualization, magnification and illumination offered by the surgical microscope, the osteotomy became more conservative, and our knowledge of the apical anatomical details has increased [1]. Entities such as isthmi, lateral canals and microfractures can now be clearly visualized. Root-end preparation involves preparing a class I cavity at least 3 mm into root dentin, with walls parallel to and within the anatomic outline of the root canal space [4]. Modern ultrasonic tips can facilitate the preparation of a 4-mm, 5-mm, 6-mm, or even longer root-end cavity [1]. Those tips are very efficient at preparing a class I cavity coaxial to the canal, even in canals with calcification or even obliteration. At the same time, root end sealing is now performed with biocompatible root end filling materials that have antibacterial properties, are dimensional stable, hydrophilic and possess a high sealing ability. Clinically, modern root end filling materials are available in a premixed form and are easy to handle. Mineral trioxide aggregate (MTA; ProRoot MTA; Dentsply, Tulsa, OK, USA) is the material of choice, and more recently bioceramic root repair materials were introduced showing promising results [5–7].
The bone window technique was first described by Khoury and Hensher in 1987 [8]. It involves the buccal approach to the apical area through a bony window. The osteotomy is precise and selectively using piezoelectric saws, without sacrificing healthy bone. Excellent exposure to the operative field and preservation of the cortical bone are obtained. The bony window is carefully preserved and after root resection and reverse filling with biocompatible materials, is repositioned to its initial place.
The technique aims to preserve more bony structure and to maintain the integrity of the buccal cortical plate. Furthermore, it serves as an autologous graft material which can provide optimal healing without the need of additional alternative regenerative materials [9]. As a result, tissue damage and complications are decreased. The bone is cut to a size so that it contains the lesion and the apical thirds of the roots and therefore provides an excellent exposure of the operation field. A necessary condition is that the cortical plate is intact. Care is taken so that the bony window is placed firmly in its initial place after the end of the surgery to avoid its penetration into the osteotomy site [10, 11].
Traditional surgical methods utilize surgical burs to perform osteotomy. However, drilling intact bone while making osteotomy results in greater bone loss and delayed healing [2, 12, 3]. On the contrary, removal of the cortical plate with piezoelectric instruments and reposition after the procedure enables adequate access to the surgical area, excellent visibility, minimal loss of bone structure and protection of special anatomical entities such as the inferior alveolar nerve. Piezoelectric devices enable a safe, selective, and precise surgical bone cut as piezoelectric function stops when the piezoelectric saw contacts soft tissue [11, 13, 14]. It is therefore a safe, predictable, and effective tool for creating and elevating the bone window. In addition, the surgery area is bloodless with great intraoperative visibility.
This study reports two cases where bone window technique was used on mandibular posterior teeth and provides a review of bone window osteotomy along with the modern microsurgical concepts and materials.
CASE REPORTS
CASE 1 (TOOTH #34)
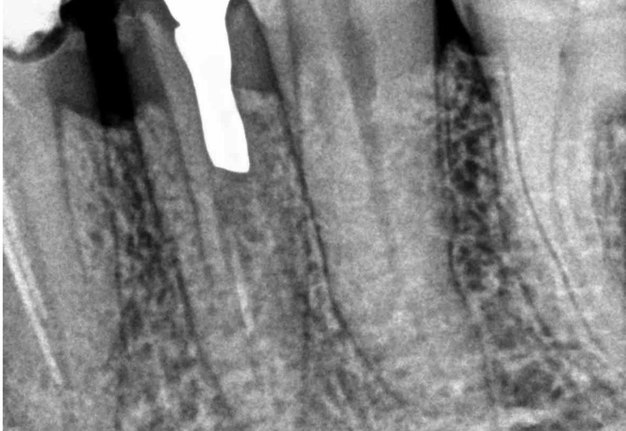
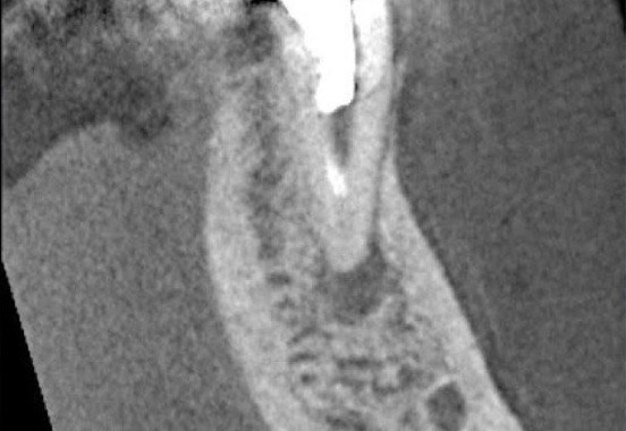
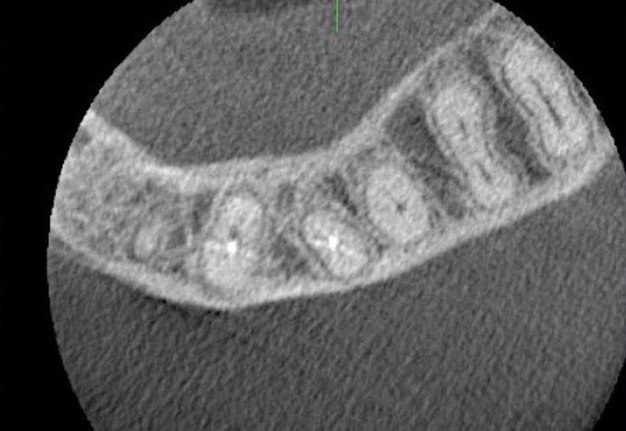
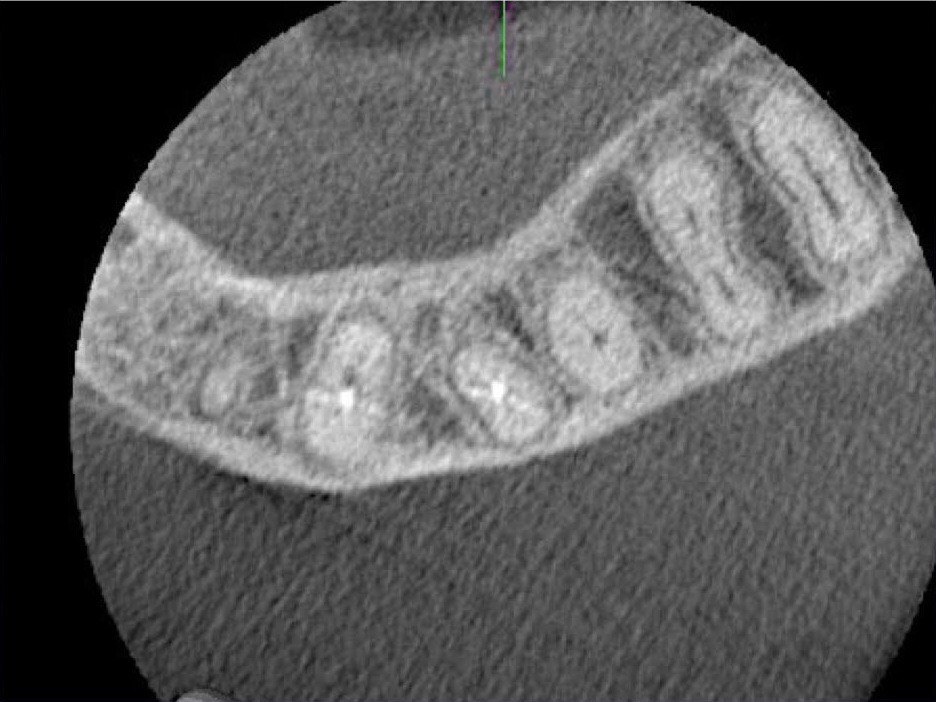
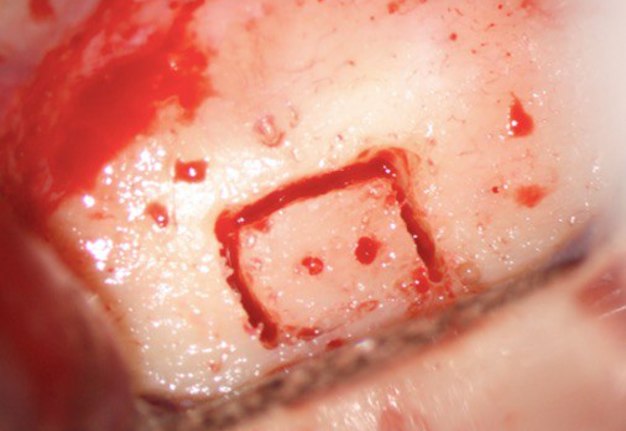
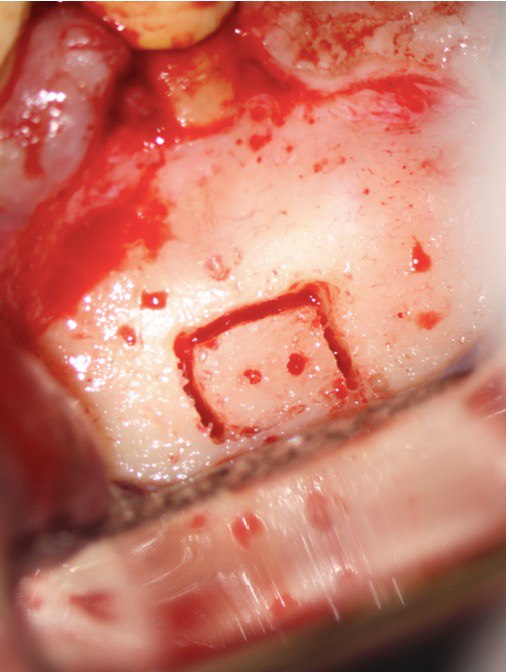
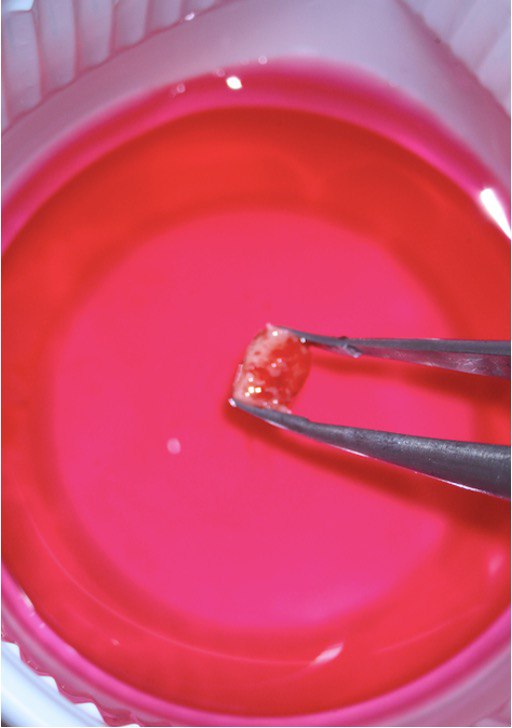
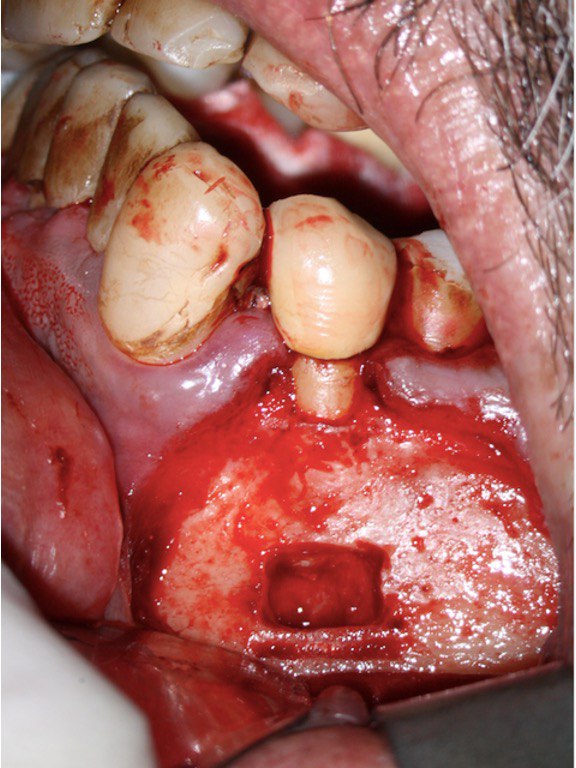
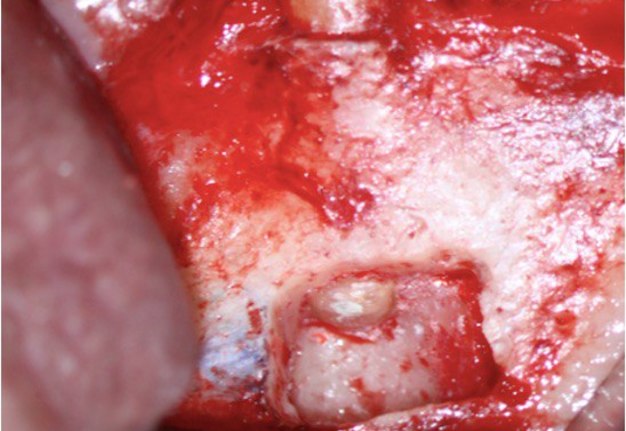
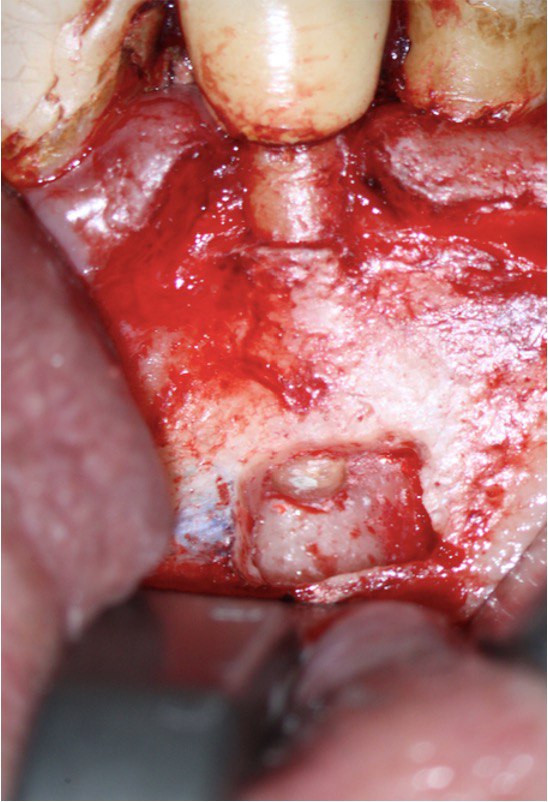
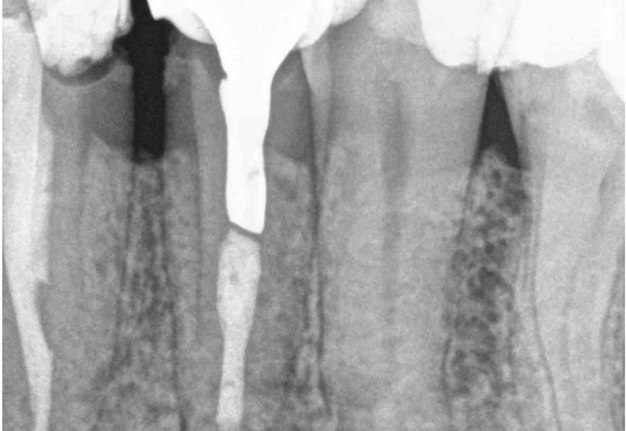
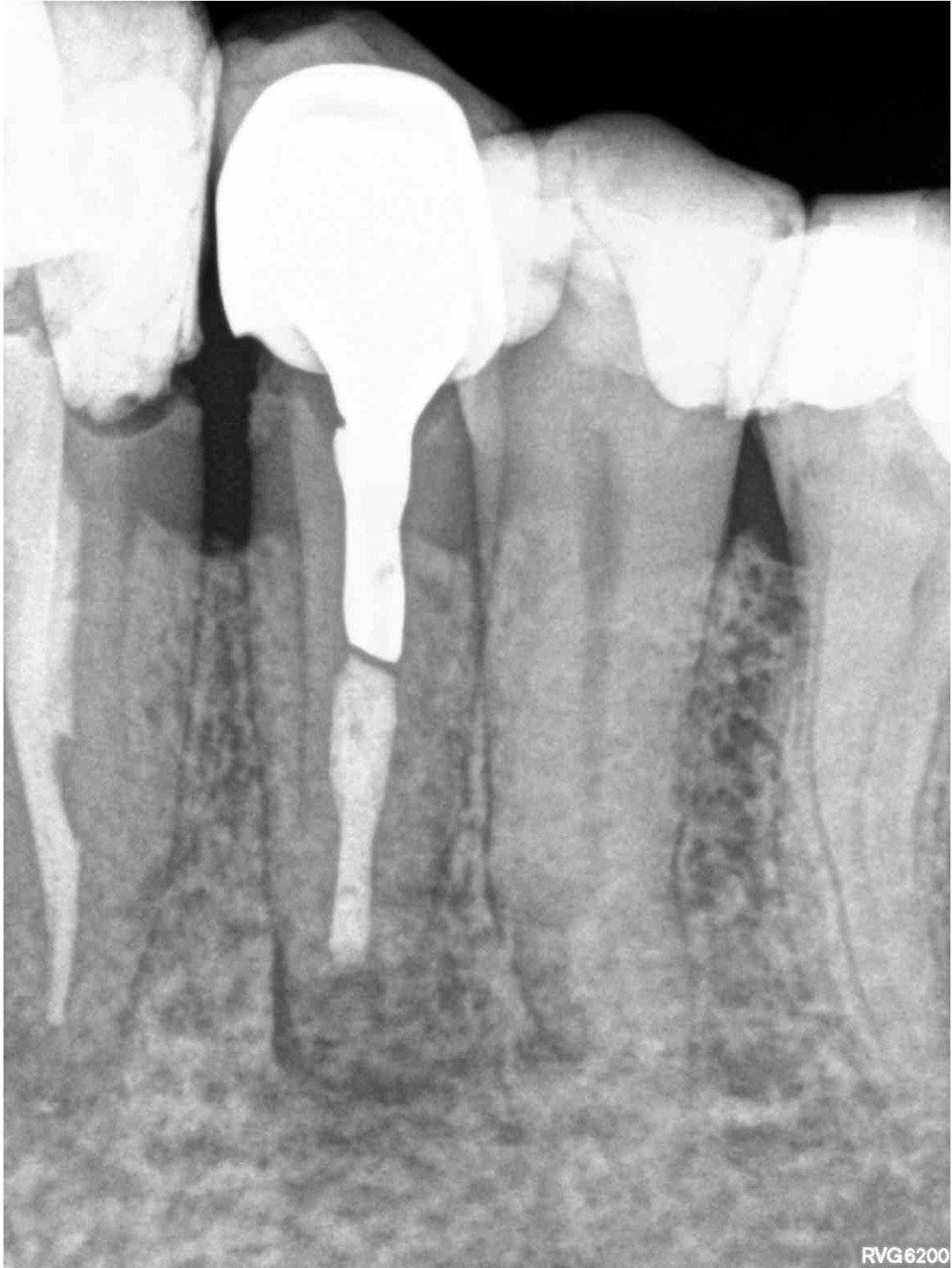
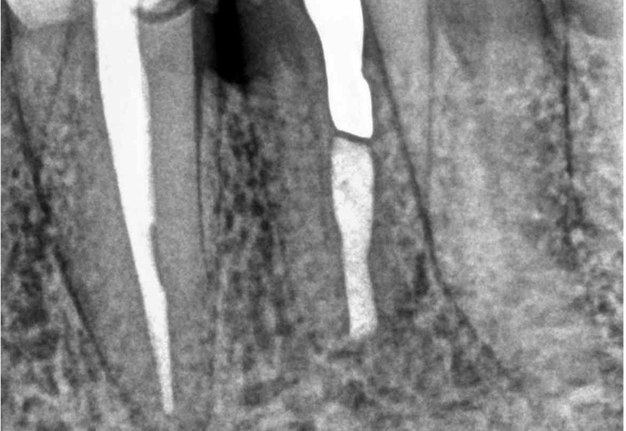
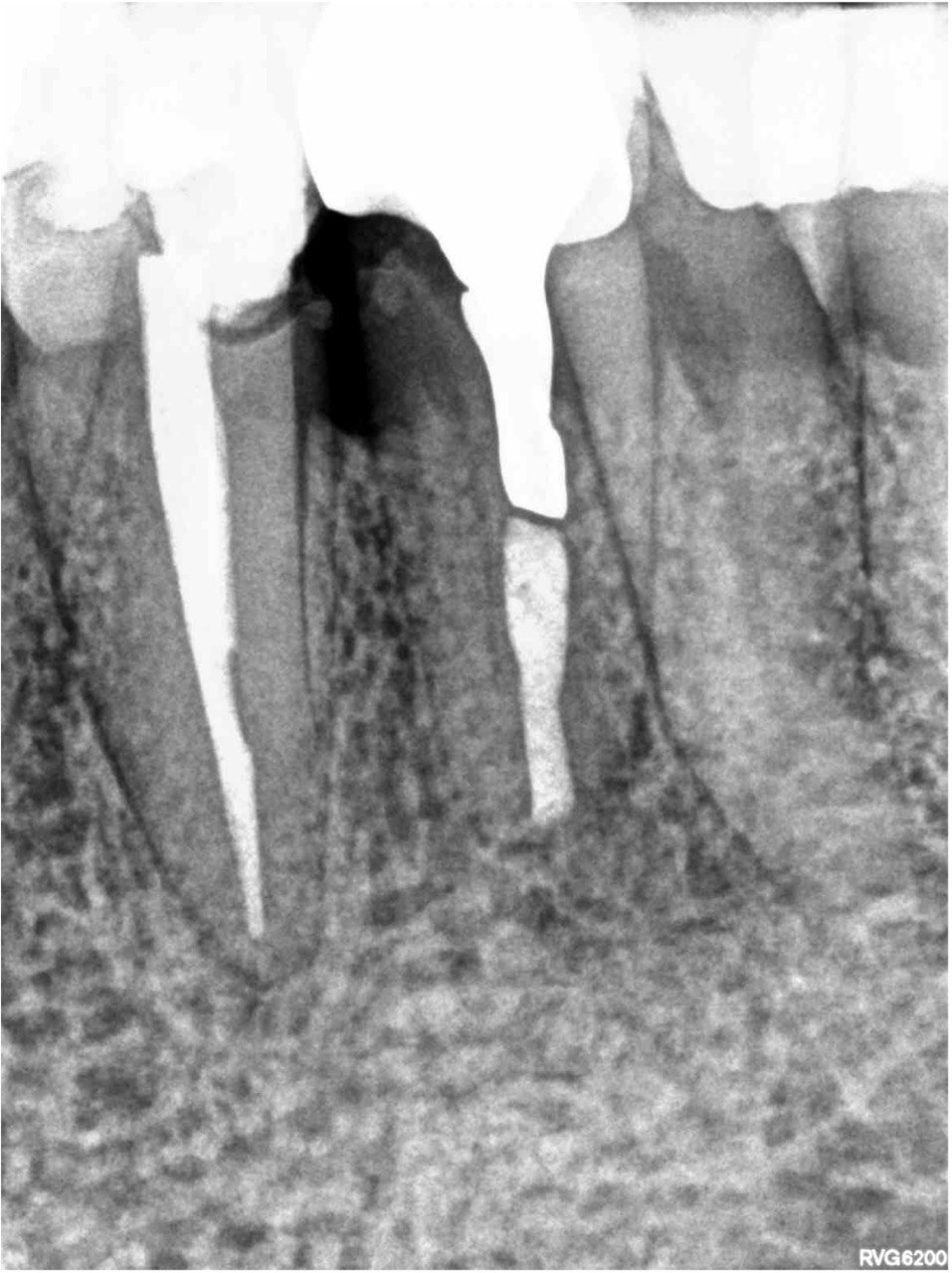
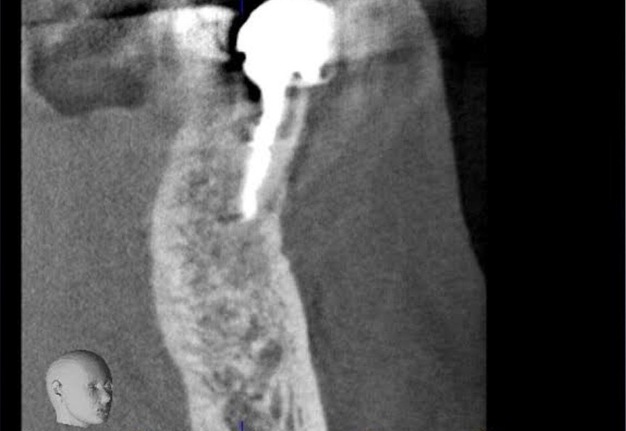
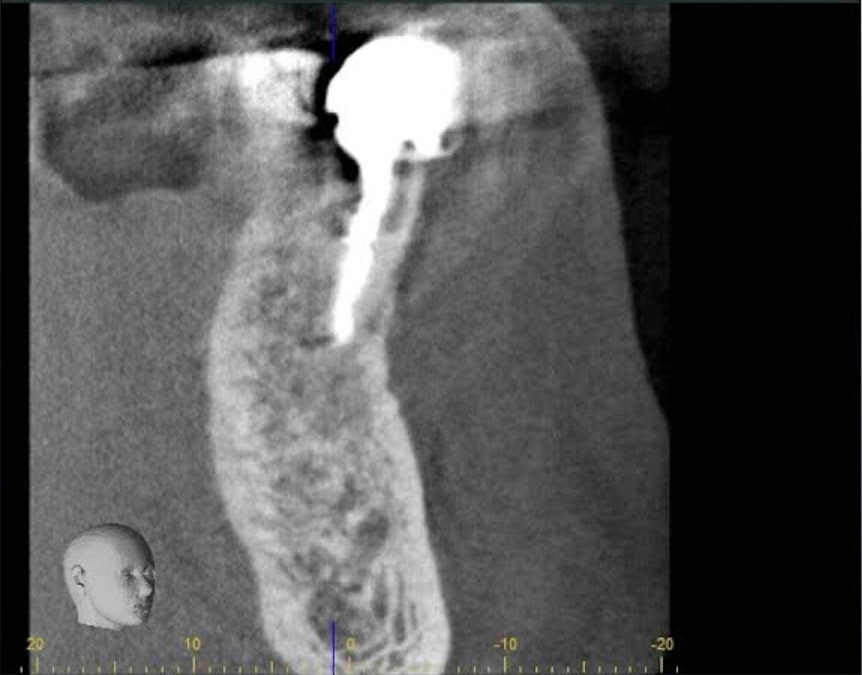
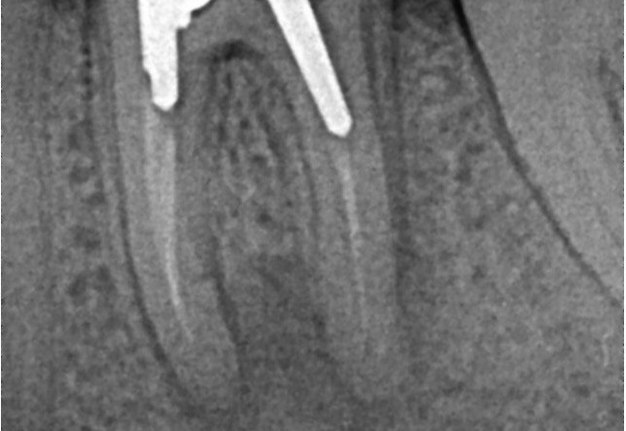
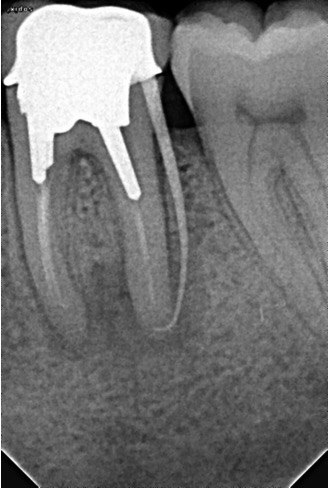
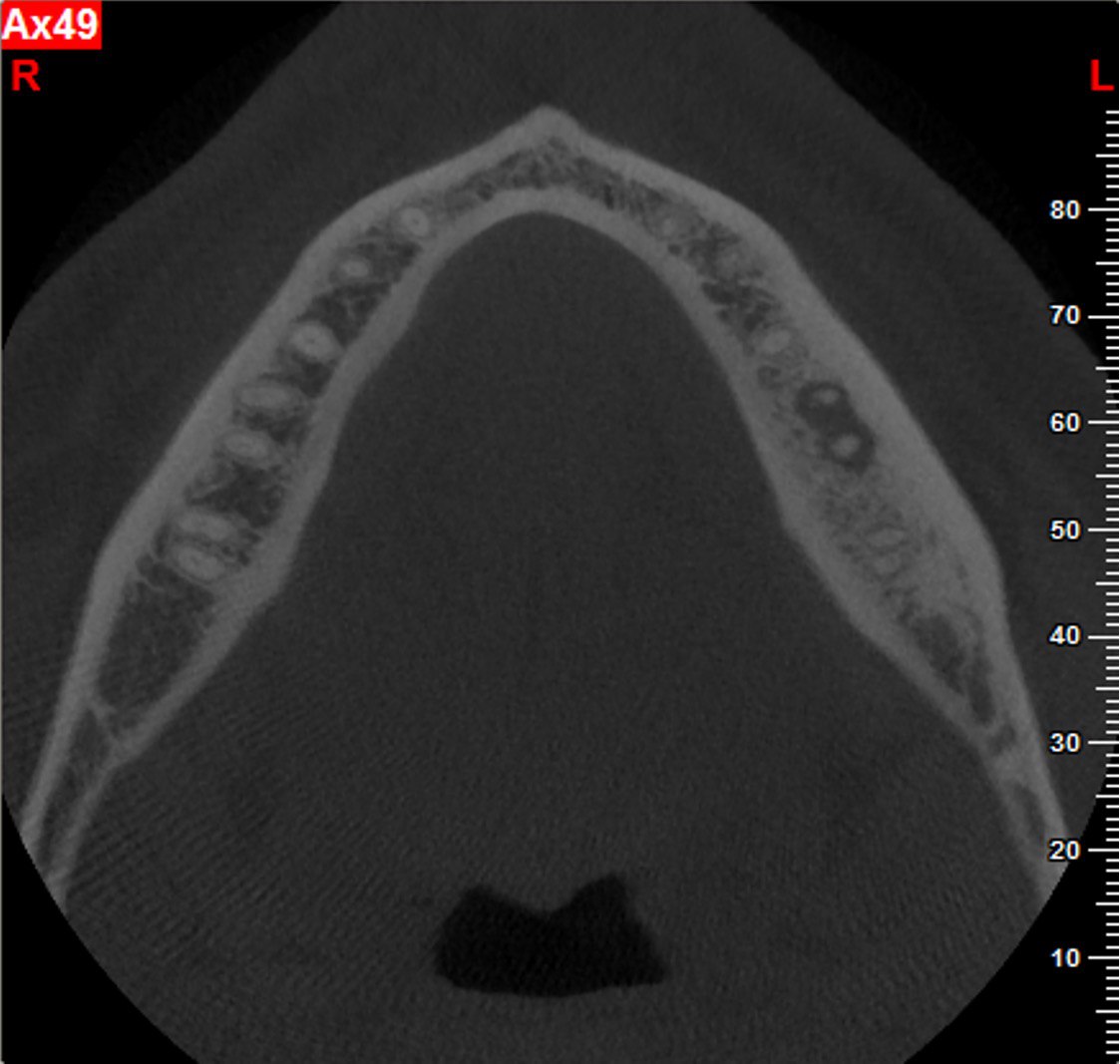
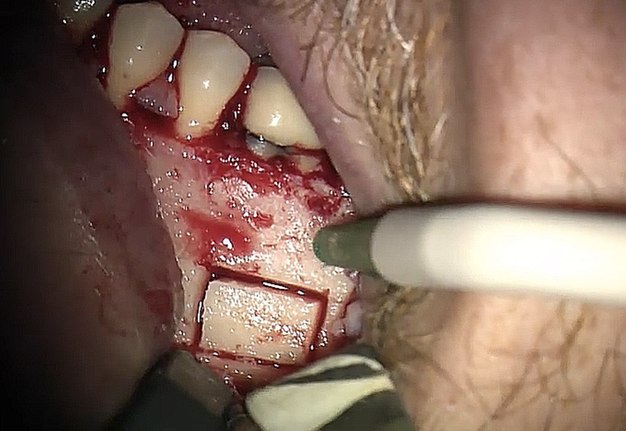
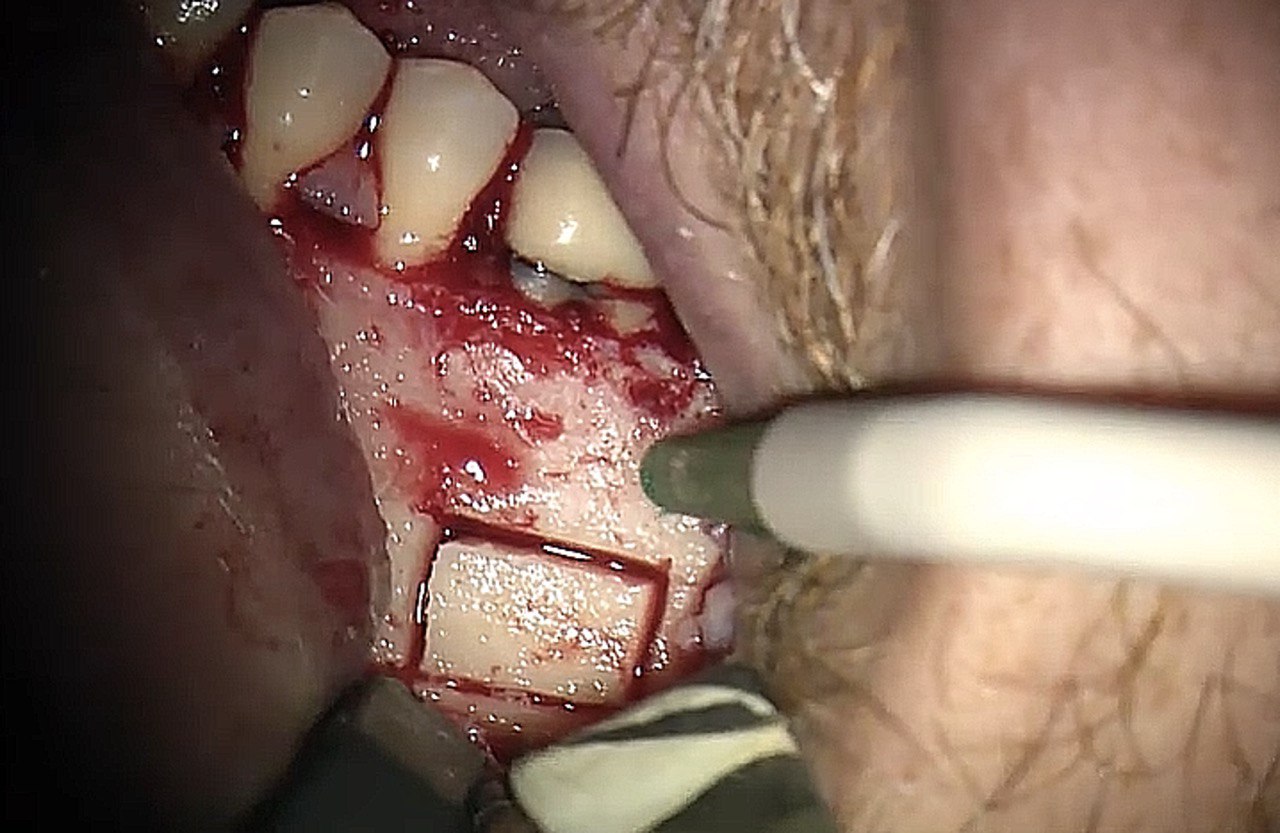
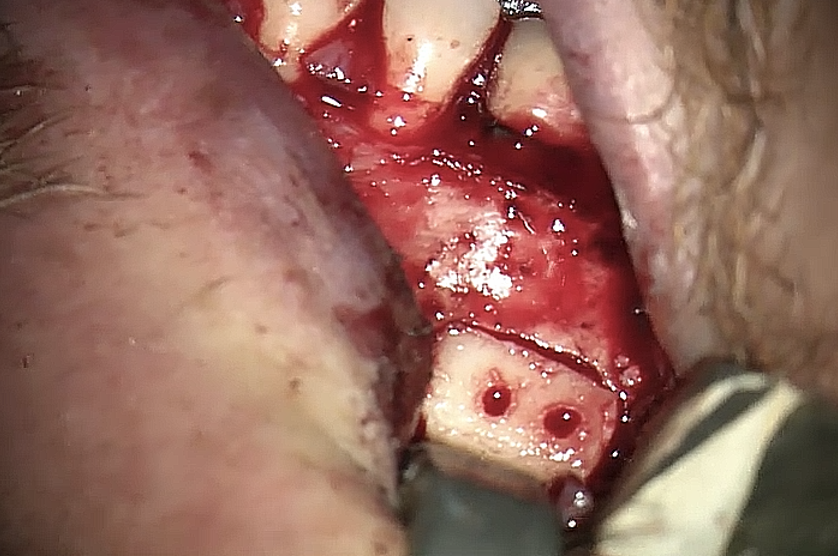
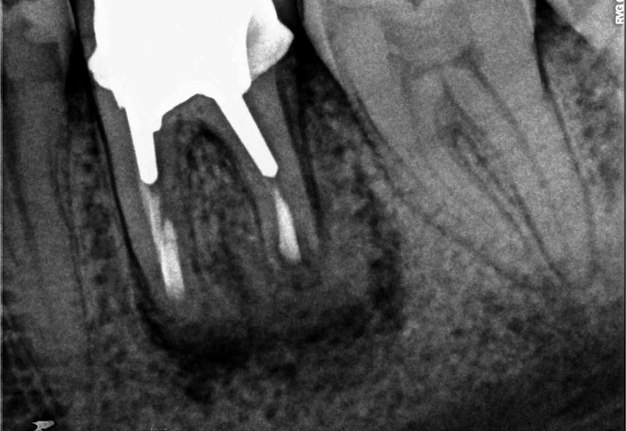
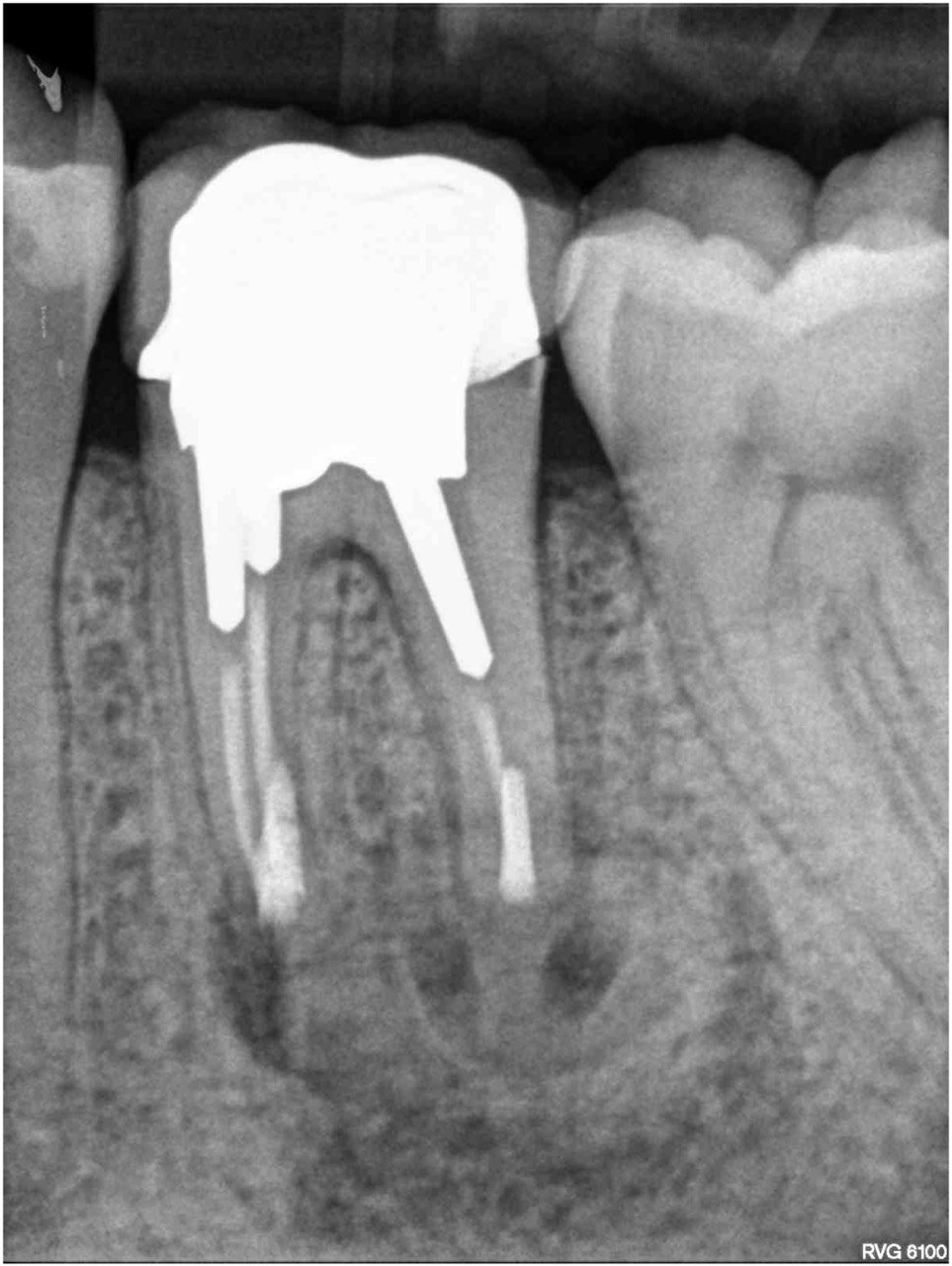
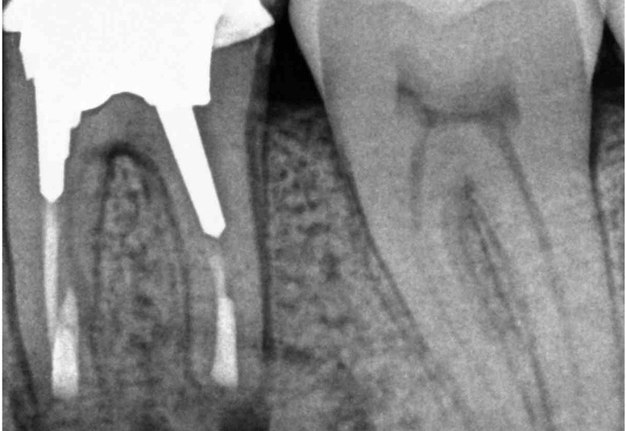
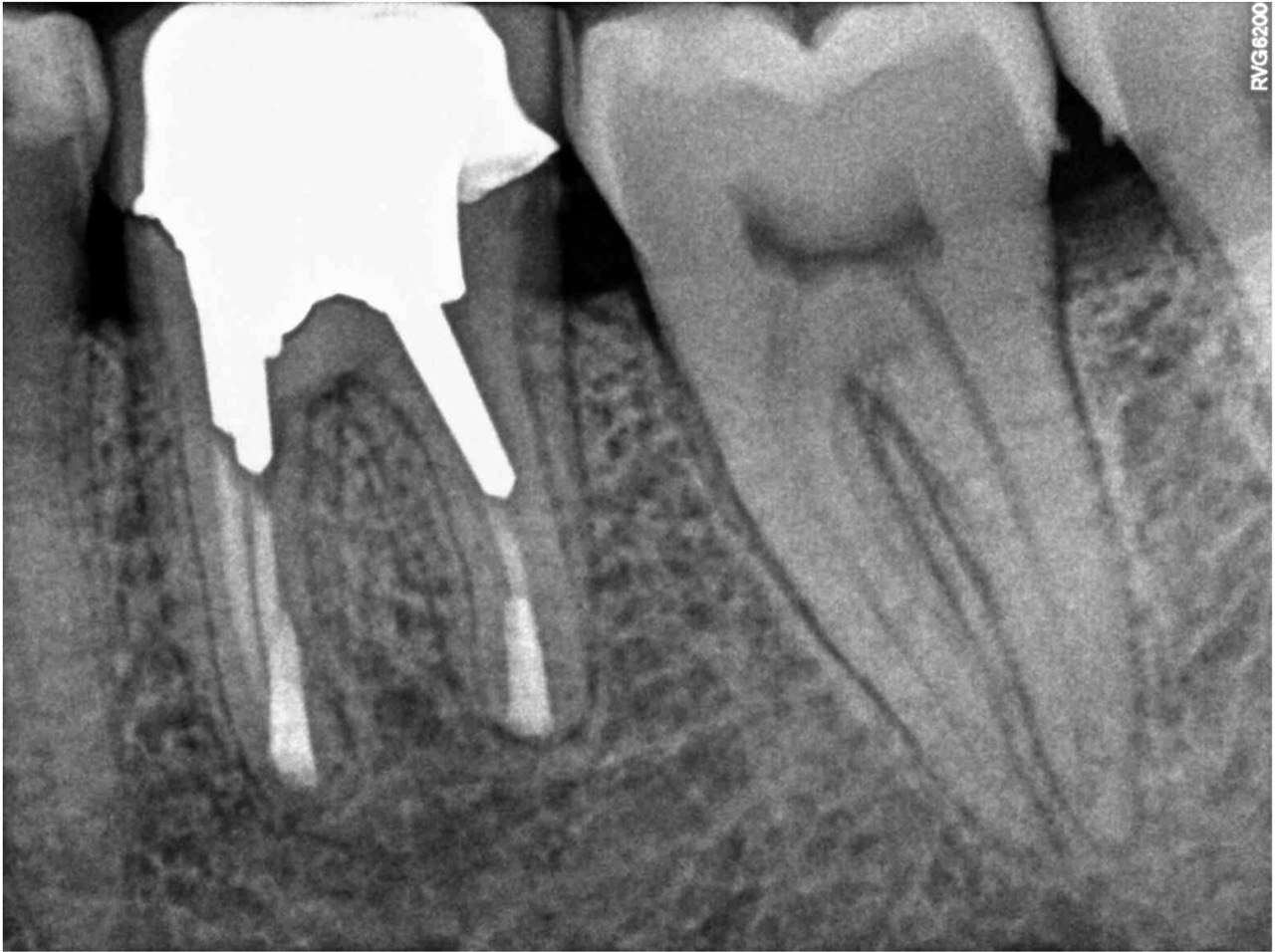
A 60-year-old male patient was referred to the private office for evaluation and treatment of a mandibular first premolar (tooth #34). He reported swelling in the area one month before his appointment. His medical history was noncontributory. In his dental history, tooth #34 was endodontically treated and restored 15 years ago. Clinical examination revealed moderate pain on percussion and palpation and a mild intraoral swelling. The tooth had post and core build up and a porcelain fused to metal crown with good margins. Periodontal probings were within normal limits. Periapical (PA) radiographs and cone-beam computed tomography (CBCT) scans were obtained and revealed a previous incomplete endodontic treatment and a 4- × 4-mm periapical radiolucency. The thickness of the buccal cortical plate was 3–5 mm (Fig 1A–C). Based on the history and clinical and radiographic examination, a diagnosis of previous endodontic treatment with symptomatic apical periodontitis was established. The patient was offered all treatment options. He opted for microsurgical retreatment. A written informed consent was given by the patient prior to surgery. After rinsing with 0.12% chlorhexidine solution (Chlorhexidine 0.12%, Chlorhexil, InterMed, Intermed S.A. Pharmaceutical Laboratories, Κifissia, Greece) for 60 seconds, the patient was administered 1 cartridge of 4% septocaine with 1:100,000 adrenaline for inferior alveolar nerve block (IANB) anesthesia and 2 capsules of 2% lidocaine with 1:50,000 adrenaline for buccal infiltration. After ensuring profound anesthesia, a full-thickness triangular flap was raised, with 4mm distal release incision, and an intact cortical plate was detected. The osteotomy was performed using a piezoelectric device (Woodpecker Surgic Touch unit, Guilin Woodpecker Medical Instrument Co. LTD, Guilin, Guangxi, China). Two vertical and two horizontal grooves were joined to create a bony window of approximately 6x6mm (Fig 1D and 1E). Bone window was removed using an elevator and the bone block was stored in HBSS (Hank’s Balanced Salt Solution, Lonza Biotech, Rome, Italy) to keep it hydrated (Fig 1F). Curettage was performed on periapical area followed by 3 mm root tip resections using a Lindeman bur under copious irrigation with sterile water. Healthy bone margins were encountered, and the root tip was clearly visible (Fig 1G). The resected root surfaces were stained with methylene blue, and inspected using a micromirror (Obtura Spartan, Fenton, MO, USA) under ×20 to ×26 magnification. An isthmus was observed joining the canals and was included in the root-end preparation. Root-end preparation was achieved using ultrasonic tips (JeTips, B&L Biotech USA Inc, Bala Cynwyd, PA, USA). The prepared root-end cavity was dried and bioceramic putty mix (TotalFill® BC RRM Putty, FKG Dentaire Sàrl, Le Crêt-du-Locle, Switzerland) was placed as a root end filling (Fig 1H). Adaptation of bioceramic to the canal was confirmed under high magnification (from ×20 to ×26). Bone window was repositioned at the original position. The flap was sutured with 5-0 monofilament sutures (Supramid nylon sutures; S. Jackson Inc, Alexandria, VA, USA) and a postoperative radiograph was taken (Fig 1I). The patient was prescribed oral analgesics (ibuprofen 600 mg 3 times a day) and instructed to rinse twice daily with a 0.2% chlorhexidine mouth rinse for one week. The sutures were removed at 7 days after surgery. The patient presented for follow-up at ten months with radiographic signs of complete healing on periapical radiograph and CBCT (Fig 1J and 1K).
FIGURE 1A. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1B. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1C. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1D. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1E. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1F. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1G. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1H. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1I. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1J. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
FIGURE 1K. Intraoral radiography showing incomplete endodontic treatment and a periapical lesion on tooth #34 (A). Limited field-of-view (FOV) preoperative CBCT, demonstrating periapical lesion of tooth #34 and an intact buccal cortical plate (B, C). Bone window is prepared. Two openings were created to facilitate blood circulation (D, E). Buccal bone placed in HBSS hypertonic solution (F). Osteotomy site after root tip resection (G) and root end filling (H). Immediate postoperative PA radiograph of tooth #34 (I) demonstrating bone fragment reposition. 10 month follow up demonstrates healing (J) and preservation of the buccal cortical plate on the coronal view of CBCT (K).
CASE 2 (TOOTH #36)
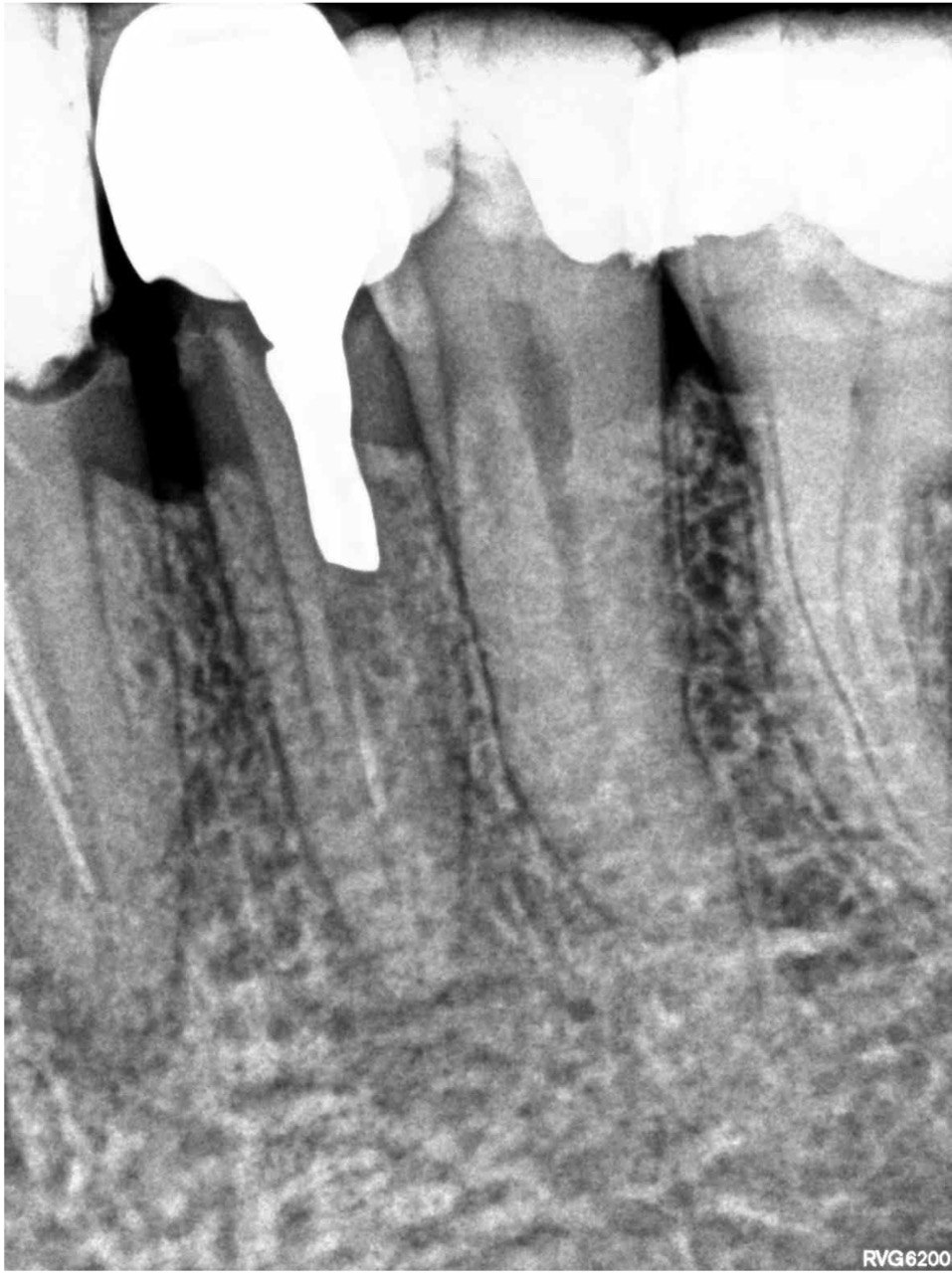
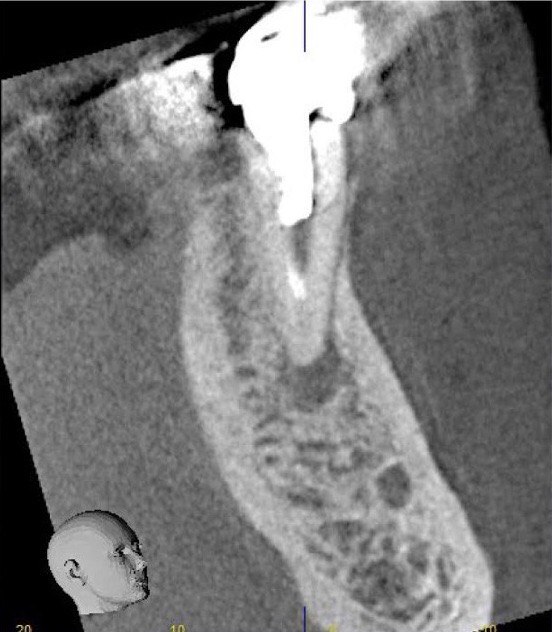
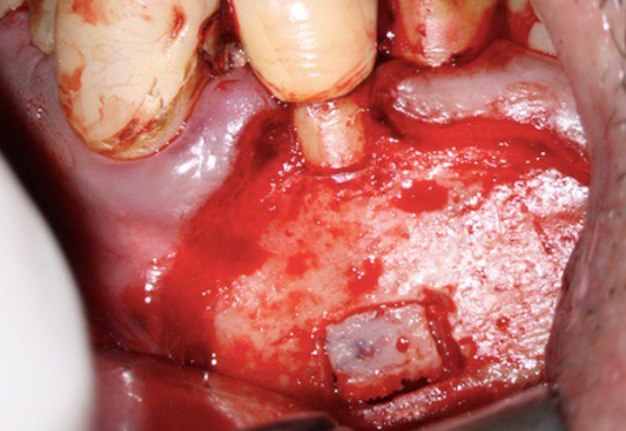
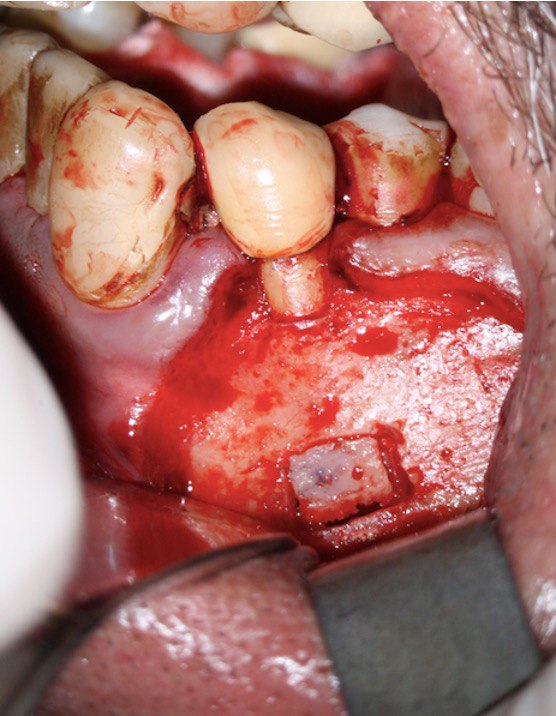
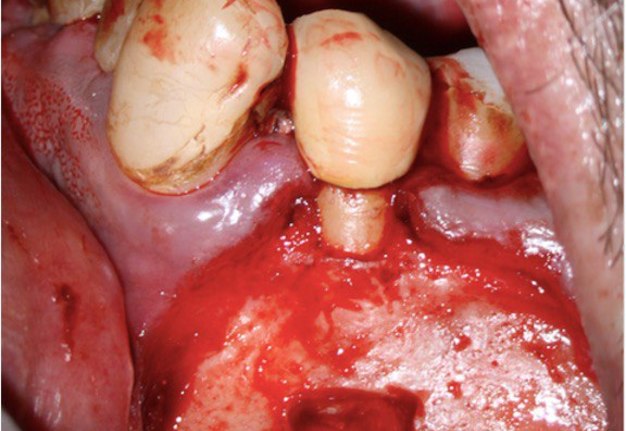
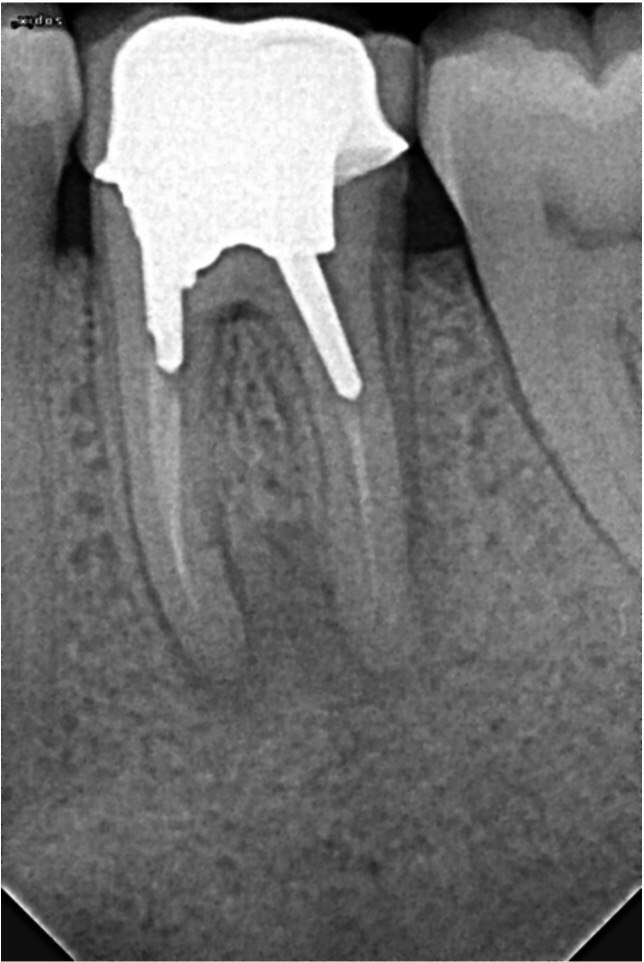
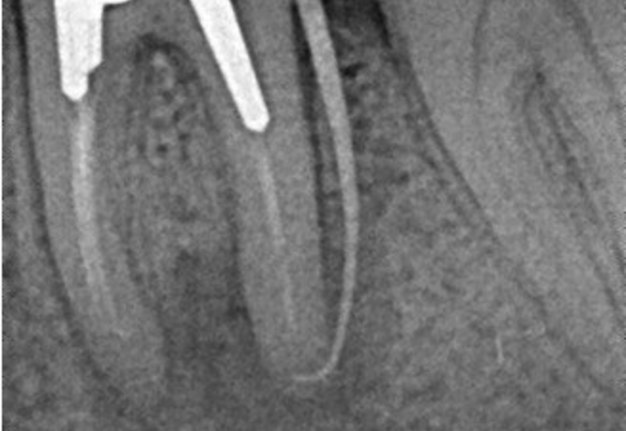
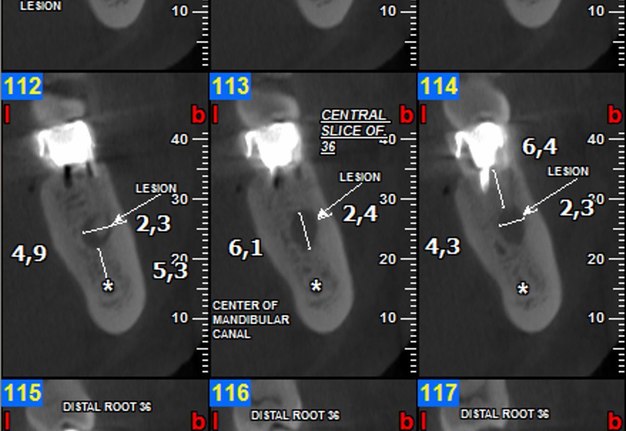
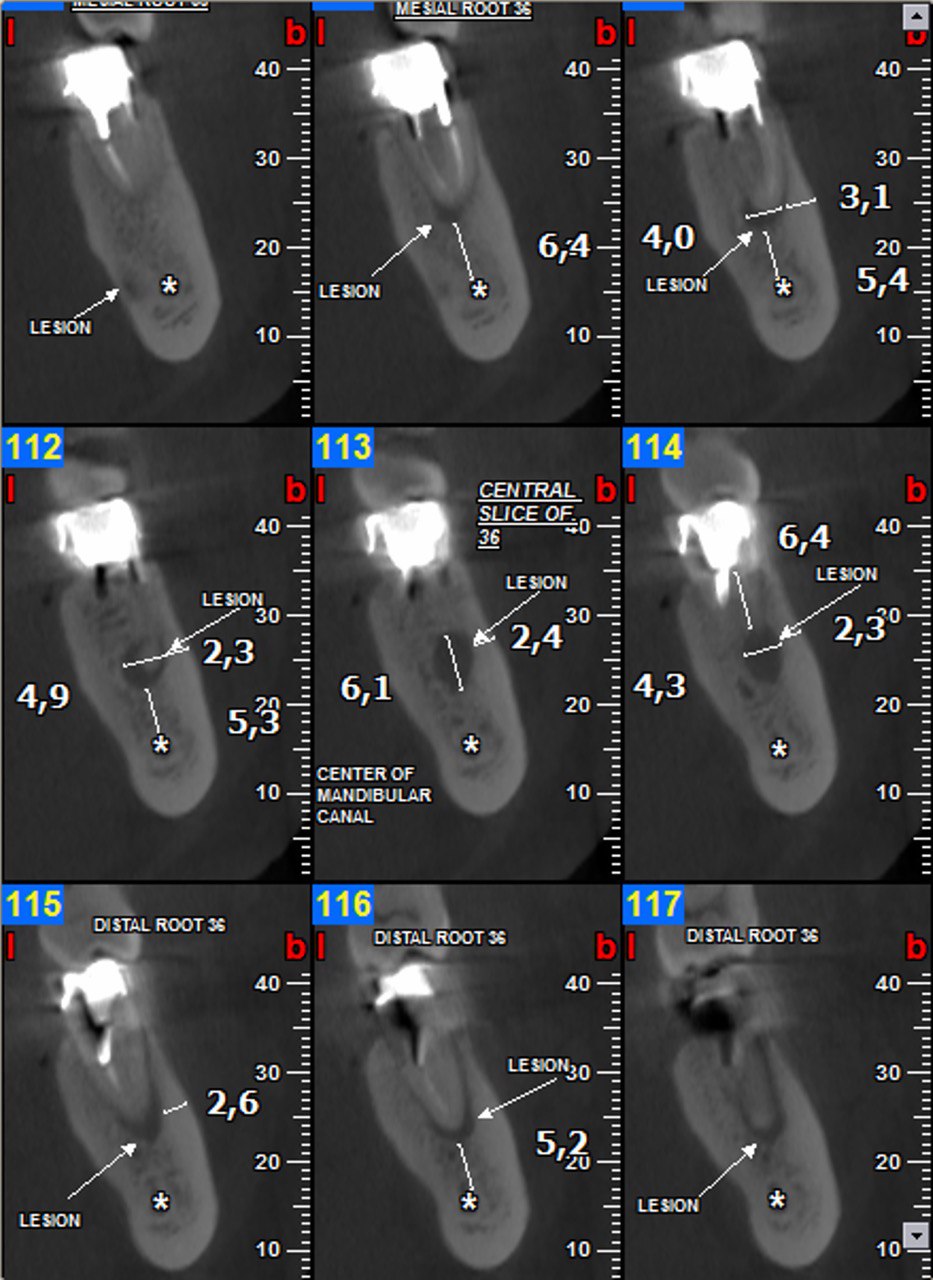
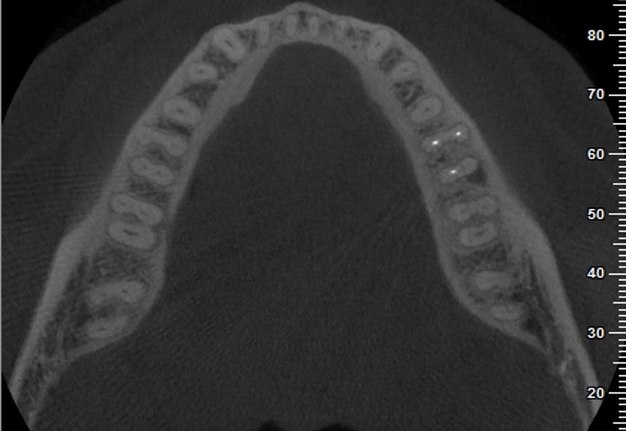
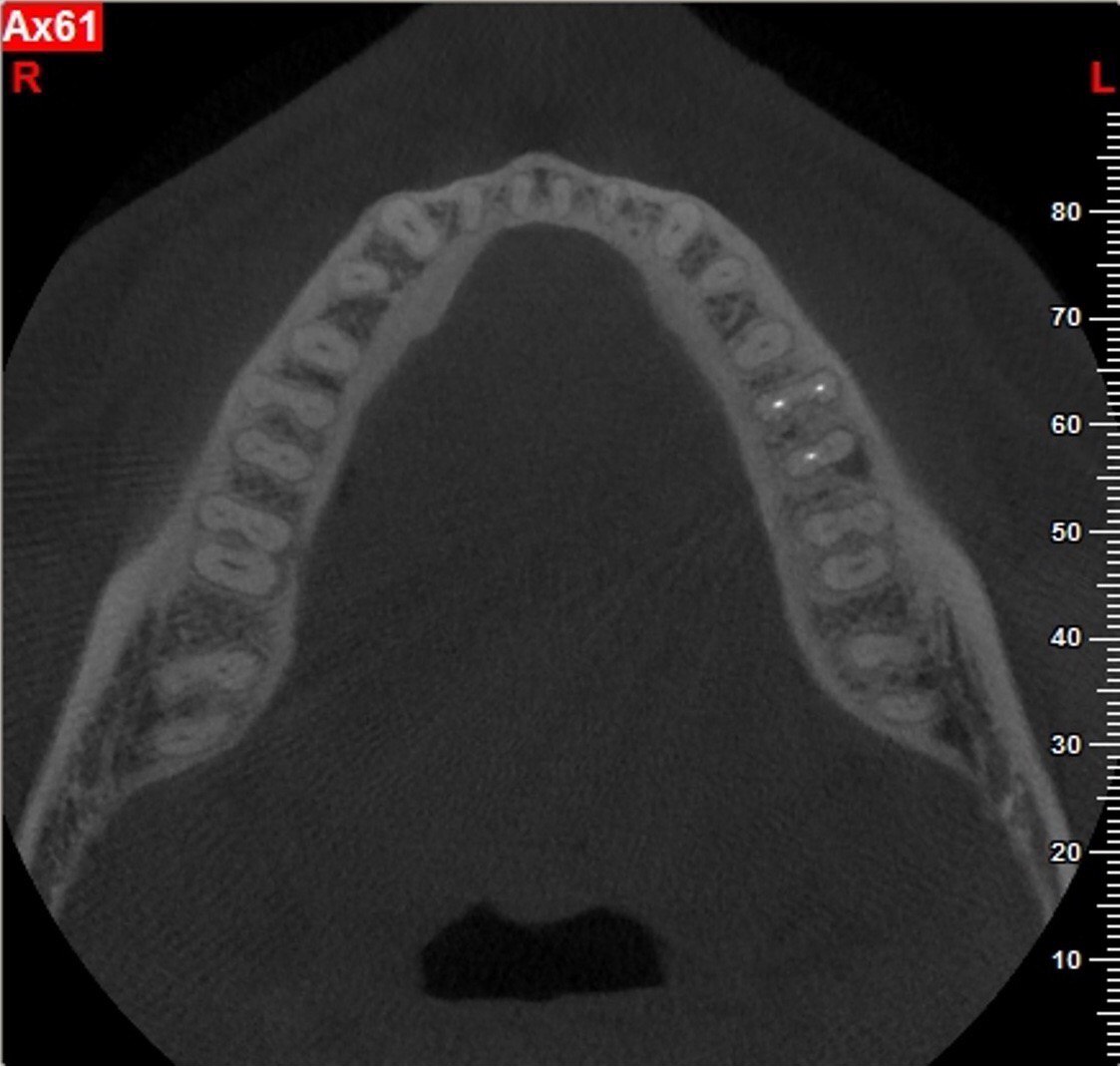
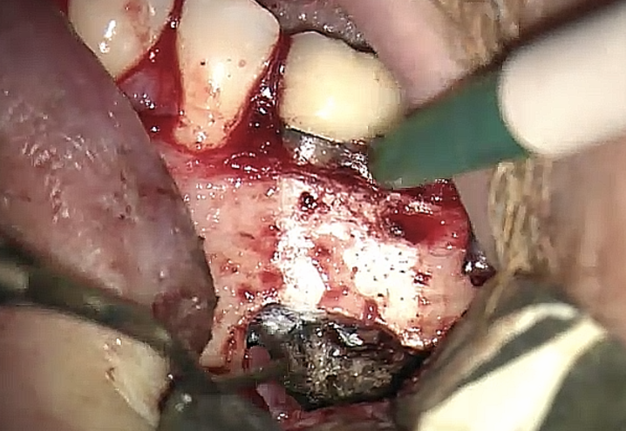
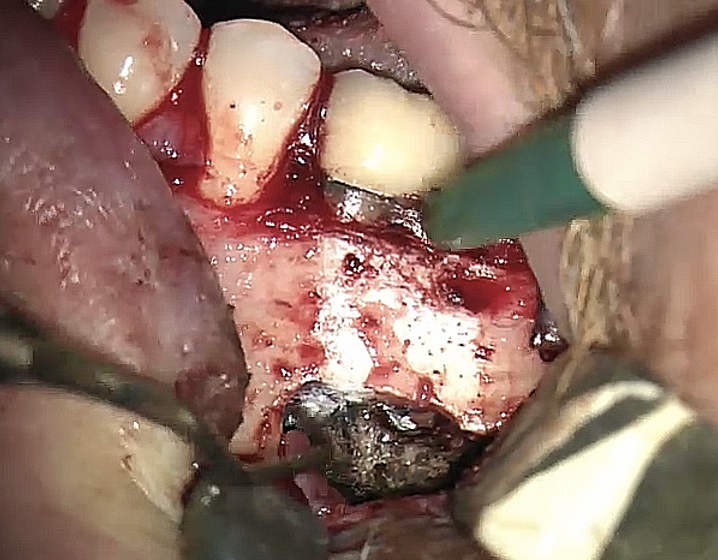
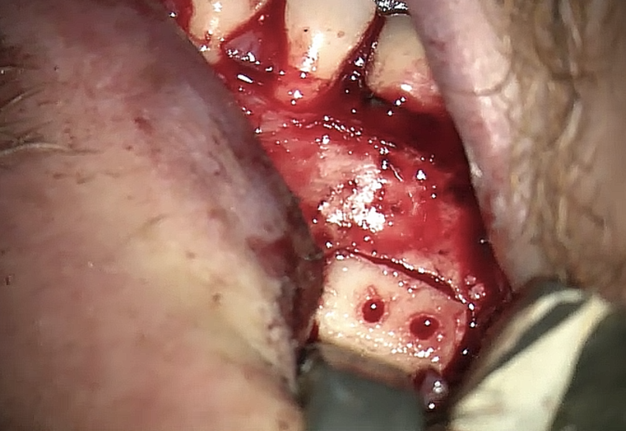
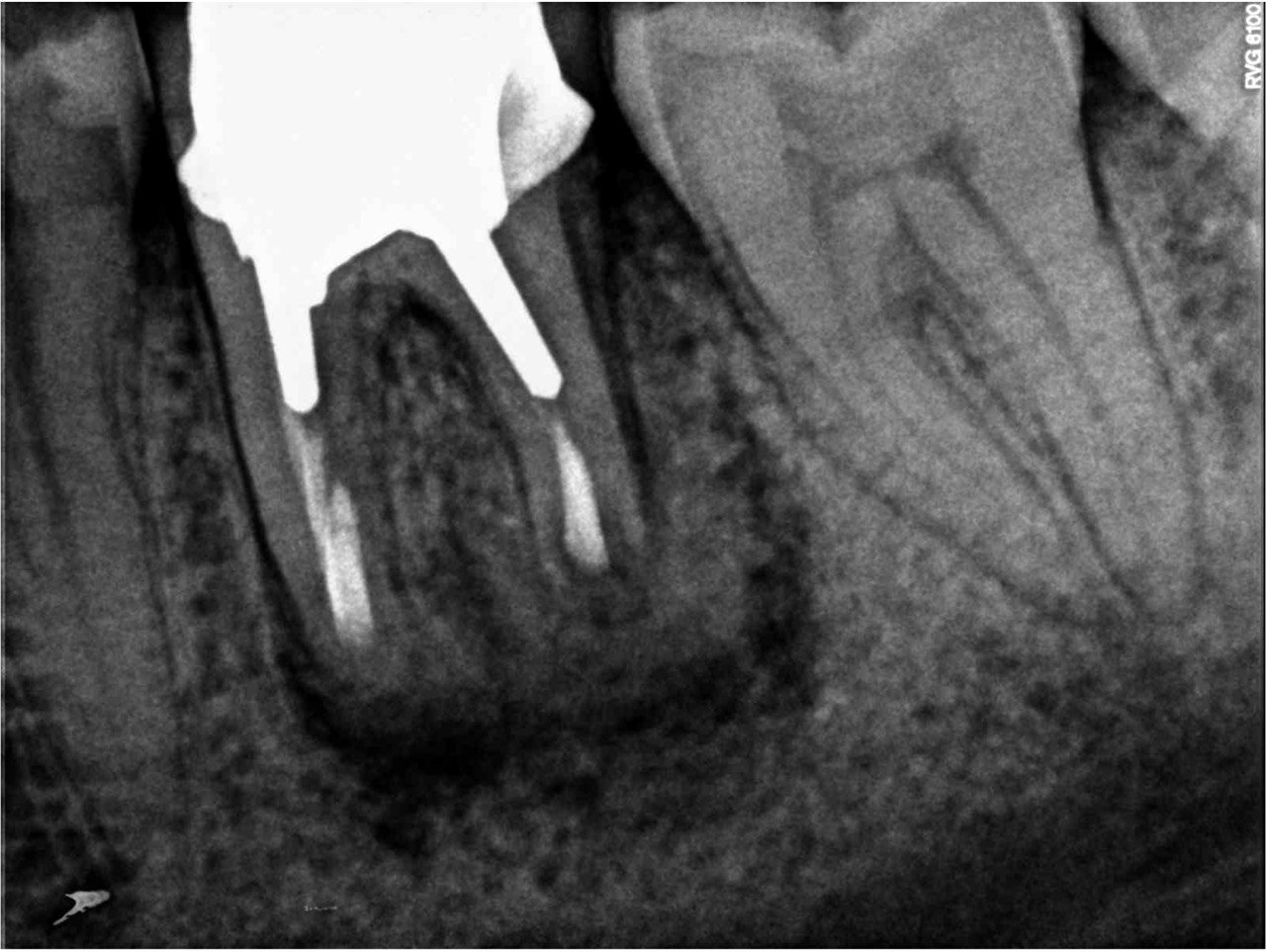
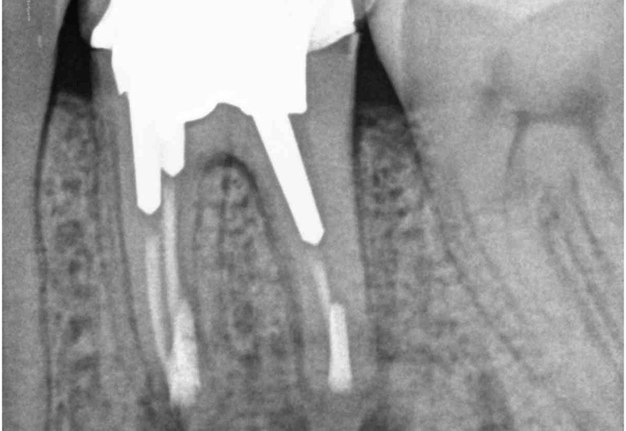
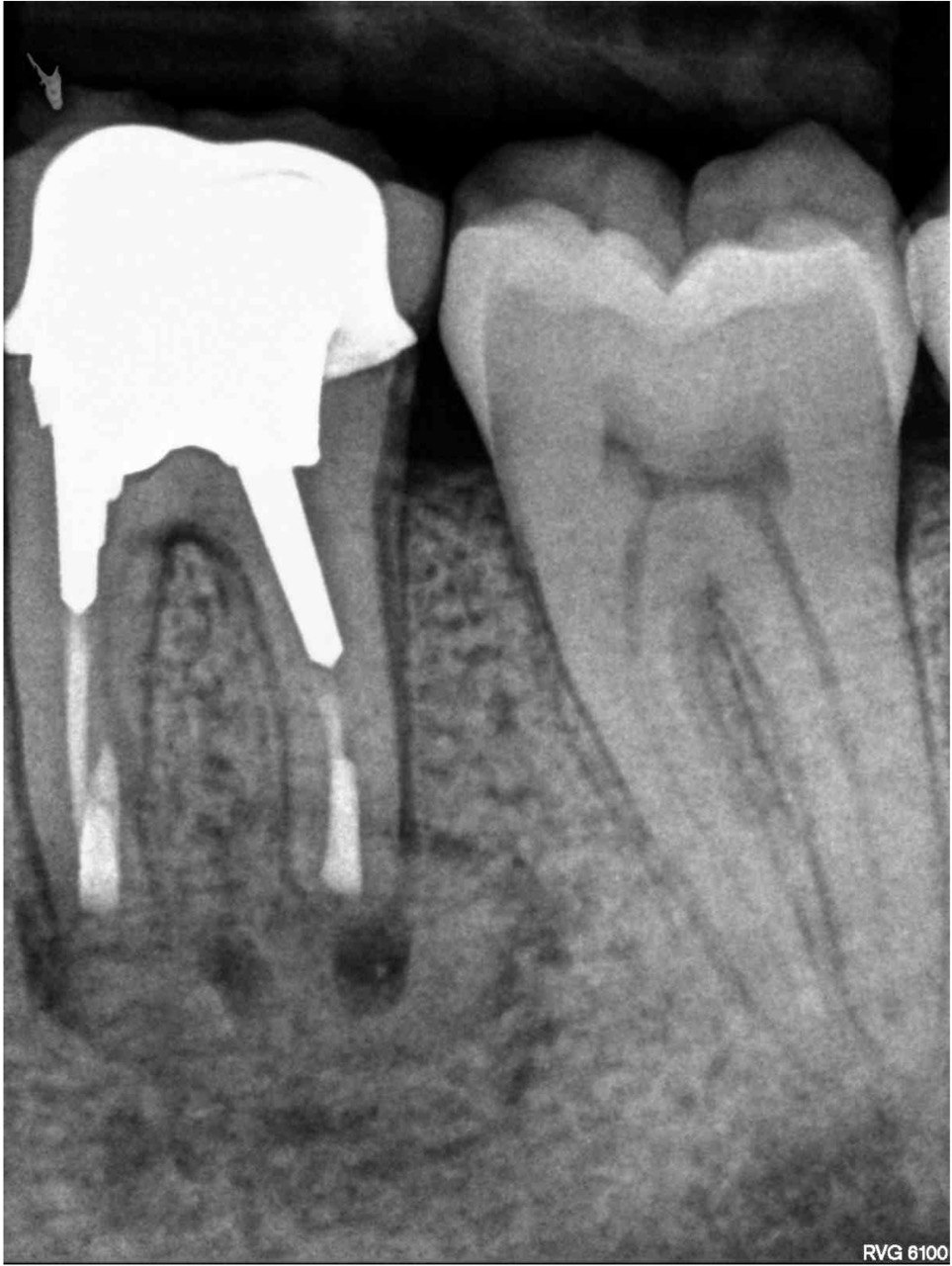
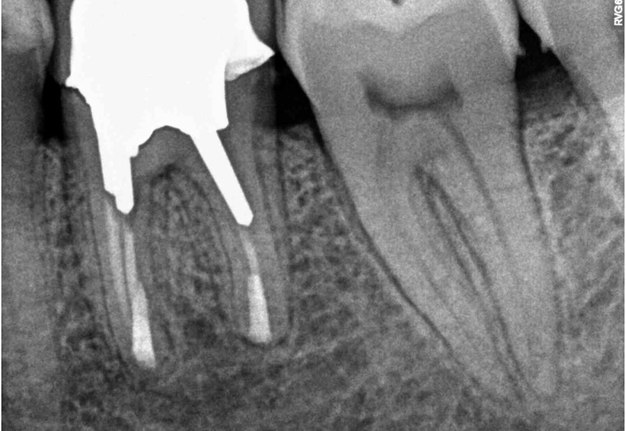
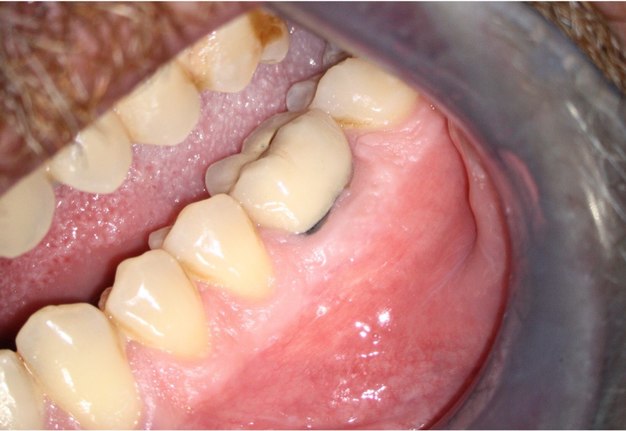
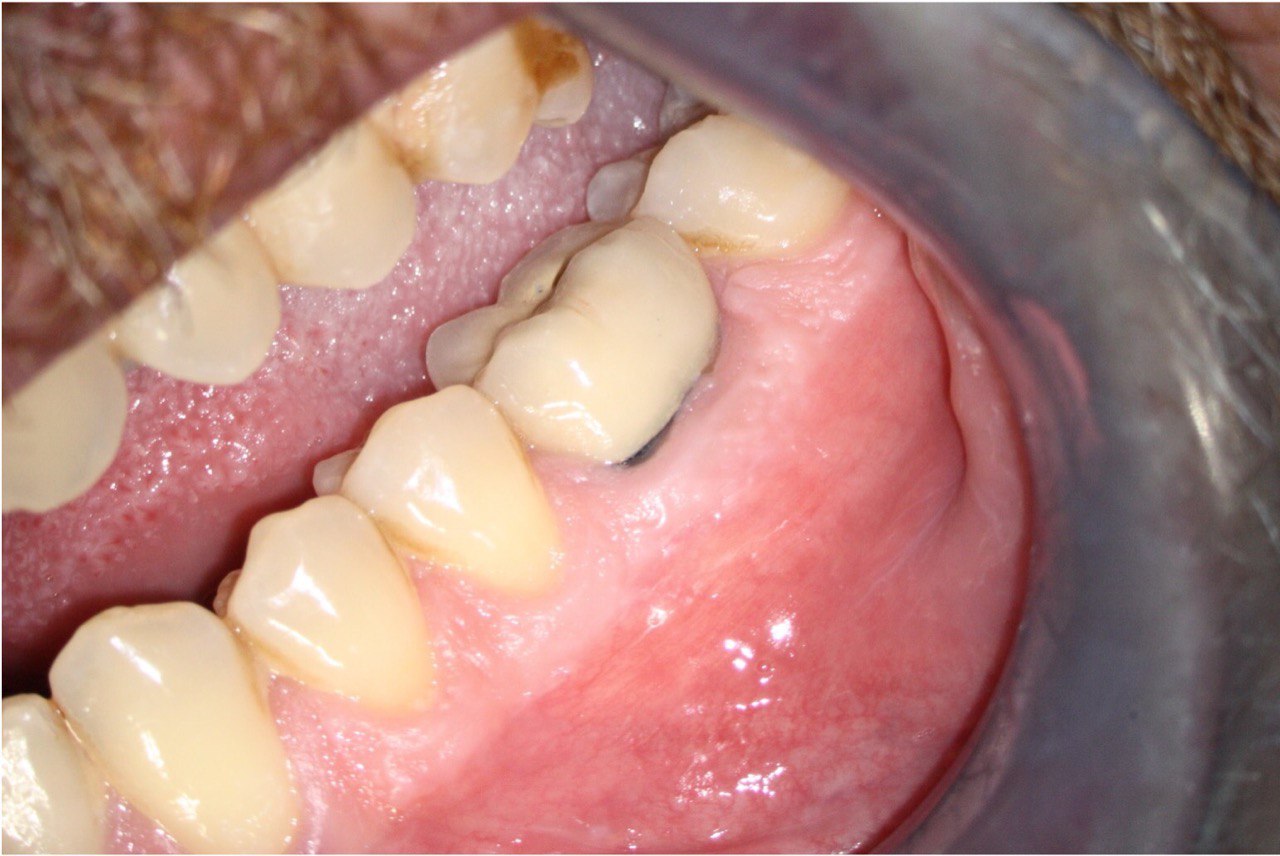
A 62-year-old male patient was referred to the private office for treatment of the mandibular first molar (tooth #36). Tooth #36 was endodontically treated 15 years ago. Medical history was noncontributory. Tooth was asymptomatic to percussion and palpation upon clinical examination. There was a buccal sinus tract tracing the apical area of #36 as confirmed by the periapical radiograph (Fig 2B). The preoperative periapical radiograph and CBCT revealed a previous endodontic treatment with a 4- × 8-mm apical radiolucency (Fig 2A–C). There was a 3-5mm thick buccal cortical plate present (Fig 2C–E). Based on the history, clinical and radiographic examination, a diagnosis of previous root canal treatment with asymptomatic apical periodontitis was established. In discussion with the patient, apicoectomy was selected as the treatment of choice and the bone window technique was implemented following the surgical protocol described earlier. The buccal bone removed was 9mm × 5mm and was placed into HBSS after removal (Fig 2F and 2G). After the bone defect was verified, root end resection and granulation tissue removal were performed. Root-end preparation was then completed (Fig 2I) with JetTips ultrasonic tips (B&L Biotech USA Inc, Bala Cynwyd, PA, USA) and sealed with EndoSequence BC RRM Putty (Brasseler, Savannah, GA, USA). Bone fragment was repositioned at the original position using a pliers. The flap was sutured with 5-0 monofilament sutures (Supramid nylon sutures; SJackson Inc, Alexandria, VA, USA). The patient presented for follow-up at 3 months (Fig 2J and 2K). At the 36 month follow up radiographic signs of healing and no clinical signs or symptoms were observed (Fig 2L and 2M).
FIGURE 2A. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2B. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2C. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2D. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2E. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2F. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2G. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2H. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2I. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2J. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2K. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2L. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
FIGURE 2M. Preoperative periapical radiographs of the tooth #36 (A, B). Sinus tract tracing the apical lesion of the tooth (B). CBCT coronal (C) and axial (D, E) view demonstrating the amount of buccal bone thickness measured as well as the proximity to anatomical structures. Intraoperative clinical pictures of case 2 (tooth #36) show: the piezoelectrically created bone window (F), root end filling (G), and bone repositioned (H). Postoperative periapical radiograph (I). Follow up after 3 months (J, K). Follow up at three years: Radiographic (L) and clinical (M) examination showing complete healing.
DISCUSSION
By use of a rotary bur for osteotomy, a significant amount of cortical bone loss is inevitable. Increased postoperative pain, delayed healing, and other complications such as nerve damage are frequently associated with conventional surgery [2, 13]. Endodontic microsurgery with bone window osteotomy is a minimally invasive procedure that offers faster healing and a better patient response [1]. The removed cortical bone is carefully replaced in its initial position and serves as an autologous graft. It promotes a complete regeneration in the surgical site as it is both osteoinductive and osteoconductive [11]. At the same time, it prevents the formation of large residual bone defects. The preservation of the cortical bone is confirmed with the use of CBCT. Additional bone grafting is not necessary and postoperative phase is more predictable and with less discomfort for the patient. It is important though, that patients are instructed not to put any digital pressure on the surgerized area, to prevent potential displacement of the bone piece.
In the reported cases, no added graft materials were used. Microsurgically treated periapical lesions can heal completely without the use of bone grafts or membranes. However, it is important to mention that guided tissue regeneration (GTR) and guided bone regeneration (GBR), when used, result in favorable healing outcomes. Bone grafts, membranes, and bioceramics have the ability to stimulate tissue regeneration. Indications in endodontic microsurgery include a large sized lesion, the need of additional stimulation of tissue regeneration, or the prevention of bone collapse [9]. The autologous bone is the reference grafting material to achieve bone repair due to its osteogenic, osteoinductive, osteoconductive and non-immunogenic properties. GTR techniques aim at preventing the surrounding connective tissue from growing into the osseous defect and therefore promote bone healing. In the reported case of Hirsh et al [11], CollaCote collagen material (Zimmer Dental, Carlsbad, CA, USA) was used between the margins of the bone window and the surrounding cortical bone. The filler material can be used as GTR material to fill a deficiency and to hold the segment in place, to prevent it from displacement or collapse into the cavity.
Preoperative CBCT provides important information of the surgical area, buccal bone and the exact position and extent of the apical pathosis [15]. The three-dimension radiographic imaging offers the ability of representative linear measurements of the width and height of the periapical lesion, evaluation of the buccal cortical plate and the anatomical structures of the surgical site. Structures such as the adjacent root tips, inferior alveolar nerve, mental foramen, and maxillary sinus should be carefully evaluated before buccal bone is piezo-electrically removed [10, 11]. Computer-assisted design and computer-assisted manufacturing (CAD/CAM) can be applied in dental surgeries including endodontic microsurgery. As shown in recent case reports, with the aid of a 3D-printed surgical template, guided minimal osteotomy is achieved and the buccal cortical plate is successfully preserved and renders the surgical procedure less traumatic [13].
The piezoelectric surgical technique offers a great advantage compared to traditional osteotomy techniques and can be applied to a variety of cases in oral and maxillofacial surgery. The unique function of the piezoelectric saw through its piezoelectric ultrasonic vibrations, offers the ability to cease its action when it comes in contact with nonmineralized tissue [16]. Therefore, it reduces the risk of accidental injuries to special anatomical structures such as the inferior alveolar nerve or the sinus membrane [17]. Due to its precise and selective cut, it differs from drilling with conventional burs that do not distinguish hard from soft tissue. Safe and minimally invasive surgeries can be conducted thanks to the minimized bone loss and preservation of the cortical plates. In addition, these thin piezoelectric saws produce less intraoperative bleeding, because of the cavitation effect of the coolant being used. Therefore, better accessibility and visibility provide the operator with precision and ease [11, 13].
CONCLUSION
Two cases were reported in this study, in which the bone window technique was predictably used. Radiographic follow up evaluation at 10 and 36 months revealed a complete healing with an intact buccal bone and no buccal indentation present.
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
REFERENCES (17)
-
Floratos S, Kim S. Modern endodontic microsurgery concepts: a clinical update. Dent Clin North Am. 2017;61(1):81-91. Crossref
-
Kim S, Kratchman S. Modern endodontic surgery concepts and practice: a review. J Endod. 2006;32(7):601-623. Crossref
-
Tsesis I, Rosen E, Taschieri S, Telishevsky Strauss Y, Ceresoli V, Del Fabbro M. Outcomes of surgical endodontic treatment performed by a modern technique: an updated meta-analysis of the literature. J Endod. 2013;39(3):332-339. Crossref
-
Carr GB. Ultrasonic root end preparation. Dent Clin North Am. 1997;41(3):541-554.
-
Rencher B, Chang AM, Fong H, Johnson JD, Paranjpe A. Comparison of the sealing ability of various bioceramic materials for endodontic surgery. Restor Dent Endod. 2021;46(3):e35. Crossref
-
Küçükkaya Eren S, Parashos P. Adaptation of mineral trioxide aggregate to dentine walls compared with other root-end filling materials: A systematic review. Aust Endod J. 2019;45(1):111-121. Crossref
-
Solanki NP, Venkappa KK, Shah NC. Biocompatibility and sealing ability of mineral trioxide aggregate and biodentine as root-end filling material: a systematic review. J Conserv Dent. 2018;21(1):10-15. Medline
-
Khoury F, Hensher R. The bony lid approach for the apical root resection of lower molars. Int J Oral Maxillofac Surg. 1987;16(2):166-170. Crossref
-
Montero-Miralles P, Ibáñez-Barranco R, Cabanillas-Balsera D, Areal-Quecuty V, Sánchez-Domínguez B, Martín-González J, Segura-Egea JJ, Jiménez-Sánchez MC. Biomaterials in periapical regeneration after microsurgical endodontics: a narrative review. J Clin Exp Dent. 2021;13(9):e935-e940. Crossref
-
Lee SM, Yu YH, Wang Y, Kim E, Kim S. The application of "bone window" technique in endodontic microsurgery. J Endod. 2020;46(6):872-880. Crossref
-
Hirsch V, Kohli MR, Kim S. Apicoectomy of maxillary anterior teeth through a piezoelectric bony-window osteotomy: two case reports introducing a new technique to preserve cortical bone. Restor Dent Endod. 2016;41(4):310-315. Crossref
-
Tsesis I, Rosen E, Tamse A, Taschieri S, Del Fabbro M. Effect of guided tissue regeneration on the outcome of surgical endodontic treatment: a systematic review and meta-analysis. J Endod. 2011;37(8):1039-1045. Crossref
-
Kim U, Kim S, Kim E. The application of "bone window technique" using piezoelectric saws and a CAD/CAM-guided surgical stent in endodontic microsurgery on a mandibular molar case. Restor Dent Endod. 2020;45(3):e27. Crossref
-
Degerliyurt K, Akar V, Denizci S, Yucel E. Bone lid technique with piezosurgery to preserve inferior alveolar nerve. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(6):e1-e5. Crossref
-
Kopacz M, Neal JJ, Suffridge C, Webb TD, Mathys J, Brooks D, Ringler G. A сlinical уvaluation of cone-beam computed tomography: implications for endodontic microsurgery. J Endod. 2021;47(6):895-901. Crossref
-
Abella F, de Ribot J, Doria G, Duran-Sindreu F, Roig M. Applications of piezoelectric surgery in endodontic surgery: a literature review. J Endod. 2014;40(3):325-332. Crossref
-
Strbac GD, Schnappauf A, Giannis K, Moritz A, Ulm C. Guided modern endodontic surgery: a novel approach for guided osteotomy and root resection. J Endod. 2017;43(3):496-501. Crossref